
Conversely, if we try and react a metal compound with a metal lower in the reactivity series, no reaction will take place. If a metal compound reacts with a metal that’s above it in the reactivity series, a displacement reaction will occur, and the more reactive metal will take the place of the less reactive metal in the compound. What use does this series have beyond ranking the reactivity of metals, though? Well, for one, it can help us predict the outcome of certain chemical reactions. The metals designated as the transition metals in the periodic table are much less reactive, and metals such as gold and platinum prop up the bottom of the series, exhibiting little in the way of chemical reaction with any everyday reagents. They’re closely followed by the marginally less reactive group two metals. Group 1 metals, the most reactive metals in the periodic table, head up the rankings. The reactivity series offers a ranking of the metals in order of their reactivity. Caesium, the most reactive metal in the periodic table, reacts extremely violently – hence why it can’t be demonstrated in a classroom! This can be compared to other common metals, such as iron and copper, which produce no reaction when dropped into water. Lithium fizzes gently, sodium fizzes vigorously, and potassium’s reaction is so energetic it bursts into a lilac flame as it zips across the water’s surface. In this demonstration, small pieces of three different metals from group 1 of the periodic table are dropped into a large bowl of water. Metals have a range of reactivities – to illustrate this, you have to look no further than the classic alkali metals in water demonstration commonly used in chemistry classes. This graphic places a selection of common metals into order of reactivity, as well as showing their reactions with air, water and steam. 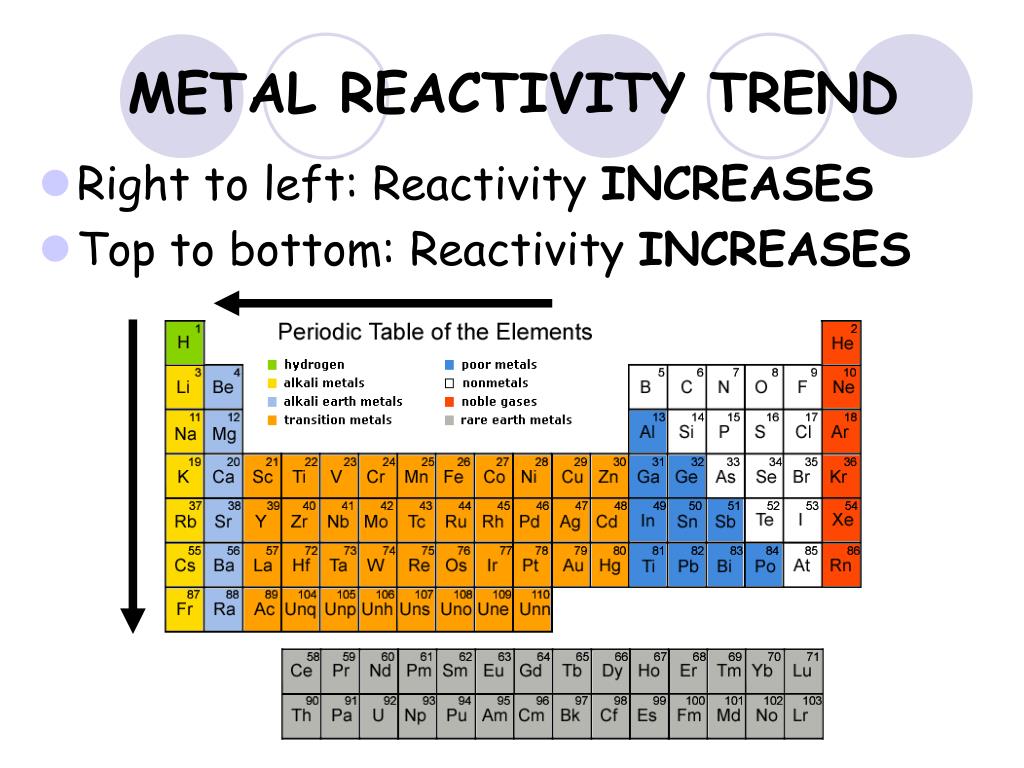
It’s also a useful tool in predicting the products of simple displacement reactions involving two different metals, as well as providing an insight into why different metals are extracted from their ores in different manners. 
The more shielding that occurs, the further the valence shell can spread out.The metal reactivity series is a commonly taught concept in chemistry, placing the metals, as its name suggests, in order of reactivity from most reactive to least reactive. The greater the number of electron shells, greater the distance between the valence electrons and the nucleus, so there is less attraction between the two. Shielding effect: electrons can shield themselves from the pull of the nucleus. Hence, francium is the most reactive Group 1 element as it has the greatest number of electron shells. Since the pull of the electron towards the nucleus decreases, the element becomes more reactive as it can more easily give up it’s electron to achieve an octet structure. As the number of shells of an atom increases, there is less attraction between the outermost valence electron and the nucleus due to more shielding. Group 1 metals are generally quite reactive as they readily give up this one valence electron to achieve a stable octet.įrancium is the last element of Group 1, which means it also has the greatest number of electron shells. 
Elements are always trying to achieve an octet configuration, which means 8 electrons in their outer shell. Group 1 elements have only one electron in their outer shell, which is the electron given up to take part in various chemical reactions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
